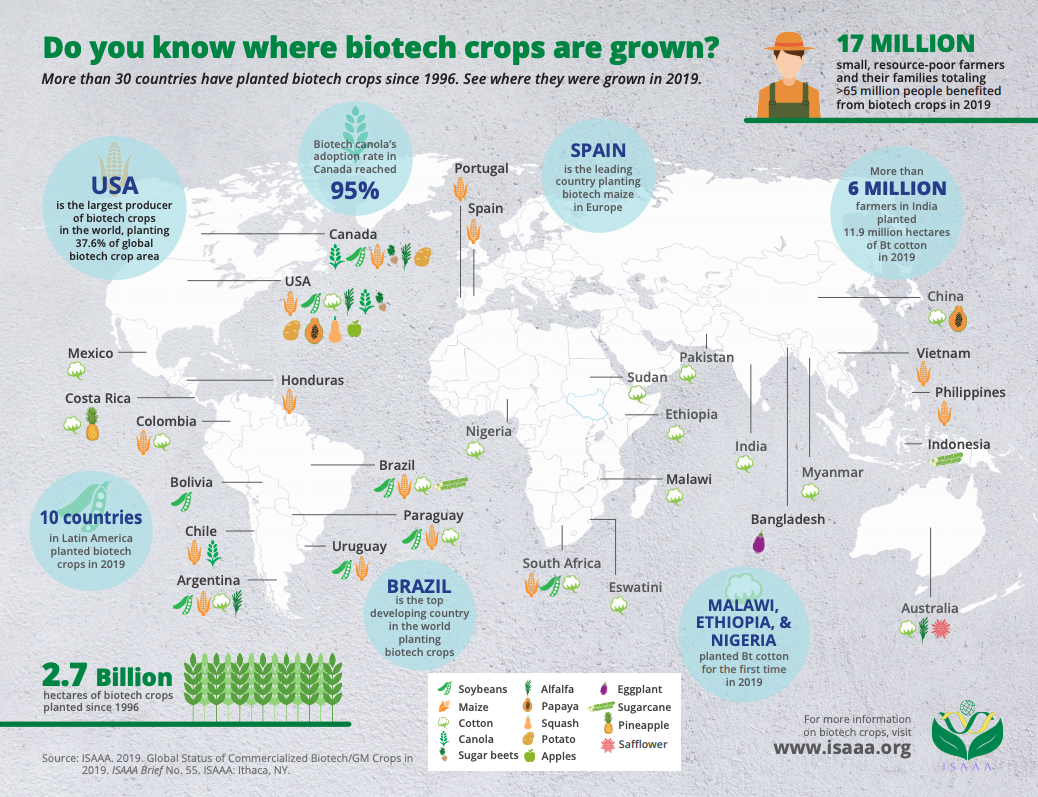
GMOs Around the World

GM crops are grown around the world by approximately 17 million farmers, most of them in developing countries. In total, more than 70 countries import or grow GMOs, and in 2019, 29 countries (five industrial and 24 developing) planted biotech crops. As of 2019, the top five countries growing GMOs in terms of crop area are the United States, Brazil, Argentina, Canada and India.
Cultivating GM crops has provided significant benefits to farmers globally, including increased yield and lower production costs. Importantly, GMOs also help to alleviate poverty for the millions of resource-poor farmers and farm families around the world (equaling approximately 65 million people total). PG Economics estimates that farmers in developing countries received $4.41 for each dollar invested in genetically engineered crop seeds in 2018. As countries look to expand their domestic GM product pipelines and crop production, even more farmers will have access to improved seeds and the benefits they provide.
Here is a guide to where GMOs are being grown and reviewed for approval around the world, care of ISAAA. Here are the top five (5) biotech crops across the world as well:
Click to Jump to Section
Africa | Asia | North America | Europe | Latin America | World Map || Back to Top
GMO Crops in Africa
Africa faces the dual challenges of having the highest rate of population growth compared to the rest of the world – expected to increase from 926 million to 2.2 billion by 2050 - in addition to being the continent most vulnerable to impacts from climate change. These circumstances will further burden Africa’s small-holder farmers already struggling to feed their families with the additional challenge of increasing yields by up to 80 percent to meet escalating demand. Genetically engineered crops developed to solve production problems specific to developing countries can help to improve yields, enable farmers to adopt more sustainable agricultural practices, and improve farmers’ and their families’ livelihoods. Between 2018 and 2019, Africa doubled the amount of countries planting genetically modified crops, growing from 3 to 6.
According to ISAAA, South Africa is one of the top ten developing countries to plant biotech crops, growing GM maize, soybeans and cotton on nearly 6.7 million acres in 2019. The 2019 planting area represents a trend of consistent growth for hectares of GM varieties planted in South Africa from year to year. These GM varieties represent approximately 85, 95 and 100 percent of total crop plantings, respectively.
During the same year, approximately 150,000 farmers in Sudan grew over 580000 acres of GM cotton. Eleven other countries – Burkina Faso, Ethiopia, Ghana, Kenya, Malawi, Mozambique, Nigeria, South Africa, Swaziland, Tanzania, and Uganda – have been conducting field trials on 10 crops with 16 traits related to drought-tolerance, saline-tolerance, nitrogen use efficiency, and enhanced nutrient content.
As the climate continues to warm, the IPCC estimates that rainfall variability will increase in Africa, with southern Africa and the Horn of Africa likely to experience a 10 percent decrease in annual rainfall. This is particularly significant for the millions of farmers throughout this drought-prone continent who rely on rain to irrigate their crops.
Corn is the most commonly grown crop in Africa. The TELA Maize Project is an ongoing effort to develop and commercialize transgenic drought-tolerant and insect-protected maize variety for farmers in sub-Saharan Africa. The initiative is managed through the African Agricultural Technology Foundation (AAFT) and is co-funded by the Bill and Melinda Gates Foundation and the United States Agency for International Development. The TELA project builds on the previous breeding work done in the Water Efficient Maize for Africa (WEMA) initiative, which stimulated transgenic maize research in Africa for the previous decade. WEMA and TELA researchers have utilized conventional breeding, marker assisted breeding and genetic engineering technologies to improve locally grown varieties.
Researchers are also adding, or “stacking,” insect-resistance with drought-tolerance traits so that farmers can address multiple challenges with one seed, and these seeds have become much more available in recent years. Seeds with stacked traits are available in a variety of crops for a variety of traits, with herbicide tolerant/insect resistant stacking in soybean, maize, cotton and canola as the most common commercialized options. To date, the TELA project has released 5 new varieties of maize being grown in 7 countries, resulting in greater yields compared to other varieties.
In addition to maize, research and development led by the African Agricultural Technology Foundation (AATF) is ongoing to improve other staple crops in Africa, including cowpea and rice. Cowpea (also known as field pea or black-eyed pea), according to CGIAR, is a protein-rich grain that is vital for the nutrition and livelihoods of millions of people in sub-Saharan Africa and serves as a food source for livestock. Scientists in Nigeria, Ghana and Burkina Faso are working to develop a Bt cowpea that is resistant to the pod borer insect, which can devastate up to 90 percent of a farmer’s crop.
Rice consumption in Africa has increased over the years, with rice becoming a major staple crop on the continent. Low yields experienced by some farmers may be caused by high soil salinity, insufficient water, and lack of nitrogen fertilizer. AATF, along with the International Center for Tropical Agriculture and the National Agricultural Research Systems of Ghana, Nigeria, and Uganda, are developing NEWEST rice that is stacked with nitrogen-use-efficient, water-use-efficient, and saline-tolerant traits.
Scientists are also using genetic engineering to enhance the nutritional value of some staple crops in order to address malnutrition in many developing country populations. Read more about this potentially life-saving application in our section: Improving Nutrition in the Developing World.
GMO Crops in Asia
Nine Asian and Pacific countries planted biotech crops in 2019, with the world’s number one cotton producer, India, leading Pakistan, China, Philippines, Australia, Myanmar, Vietnam, Bangladesh, and Indonesia in overall GMO production. In addition, although not a cultivator of biotech food crops, Japan has been growing the biotech blue rose since 2011. With cumulative production across the region in 2016 at nearly 48 million acres and enhanced economic benefit in 2018 at $3.7 billion, several governments are committed to further expanding biotech crop production in the future.
In India, virtually all cotton grown in the country is genetically modified. Since 2002, the adoption rate of insect-resistant cotton has increased to 94 percent with over 7 million farmers planting the crop across the country. Since the first biotech cotton plantings, the cumulative economic benefit from biotech production has been $24.3 billion, with $1.5 billion in 2015 alone.
Although the leading cotton producer globally, India has not yet grown GM crops for food. Research in this area is ongoing, however, with the government conducting field trials for GM chickpeas, rice, and tomatoes. In May 2017, India’s Ministry of the Environment Genetic Engineering Appraisal Committee concluded that the publicly funded GM mustard is safe and nutritious, bringing the crop closer to approval and commercialization for food use. A similar verdict has been reached for an eggplant GM event. Despite this regulatory approval, the government still remains indecisive on if it will clear these commodities for commercial production. India does import significant quantities of GM soy oil and canola oil for human consumption.
Pakistan is another notable cotton producer, with 95 percent of its GM cotton varieties. In 2019, over seven million acres of GM cotton were cultivated, which is a slight decrease from 2018 due to water shortages, pest infestation, and uncertainty regarding approvals. Pakistan exports its cotton to other Asian countries, including India, Indonesia, Thailand and Taiwan. The enhanced economic benefit of biotech production between 2010 and 2015 is $5.8 billion, and the increased cotton production has generated additional jobs for farm laborers, particularly benefiting women.
Like India, Pakistan has yet to adopt GM food crops for cultivation but is conducting experimental and field trial research on a number of biotech crops, including maize, wheat, rice, sugarcane, brassicas, chickpea, potato and ground nut, as well as tobacco. Support for GM food crop variety commercialization is being garnered among the Pakistani farming communities, especially after successes in the maize field trials. Pakistan is a major importer of GM soy and canola oil in addition to importing GM cotton.
While China continues to be a major importer of biotech crops globally, the Chinese government is focused on increasing its domestic product pipeline and crop production. Since 1994, China has approved 73 biotech events for food, feed and processing. In 2019, China cultivated 7.9 million acres of GM cotton and papaya. There is a 95% and 85% adoption rate of cotton and papaya, respectively, among Chines farmers. Growing insect-resistant cotton has increased average yields by 10 percent, reduced insecticide use by 60 percent, and increased farm income by $23.2 billion since 1998, $1.5 billion in 2018 alone.
A key element of China’s economic development plan is increasing its domestic research and development. China’s government has provided billions in funding to grow its biotech industry, and is conducting research on wheat, rice, maize, soybeans and peanuts. In 2016, the State Council of China announced a 5 Year Plant to commercial new strains of Bt corn and cotton as well as herbicide-tolerant soy. However, none of those products were available for farmer purchase by 2020.
Australia ranks 13th globally in biotech crops production, growing GM cotton and canola on 1.5 million acres in 2016. Since its original plantings in 1996, farmer adoption of biotech cotton has grown to 98 percent. Adoption for GM canola is at 31 percent. Australia has approved 140 biotech events. The economic benefit of growing biotech crops is estimated to be $1 billion between 1996 and 2015. Research and field trials are being conducted on several plant varieties, including bananas, barley, canola, cotton, grapes, Indian mustard, maize, papaya, perennial ryegrass, pineapple, safflower, sugarcane, tall fescue, torenia, wheat, and white clover. Biotech crop production in Australia might see and increase soon, as the State Government of South Australia has been working to finalize a bill to lift their GMO production ban for the mainland of South Australia.
Over 400,000 Filipino farmers grew 2.2 million acres of maize in 2016, accounting for 65 percent of Philippines’ maize production. Since originally planting biotech maize in 2003, the cumulative economic benefit is approximately $873 million, $87.7 million in 2018. 122 biotech events (including stacks) have been approved in the Philippines, and a number of products developed with public-private sector support are at or near the field trial stage, including Golden Rice, insect-resistant brinjal and cotton, and virus-resistant papaya.
In 2016, Myanmar grew over 740,000 acres of insect-resistant cotton, with the adoption rate at 86 percent. Over 430,000 smallholder farmers have benefitted from this crop, with enhanced farm income between 2006 and 2018 at $462 million.
Vietnam is in its fifth year of cultivating genetically modified maize. In 2019, farmers grew insect-resistant and herbicide-tolerant maize on 92,000 acres, an increase in acreage by 88% from 2018, and realized increased yields between 16.5 and 25 percent compared to non-GM varieties. Vietnam has approved 22 biotech events, and imports GMO maize, soy, cotton, canola and alfalfa. In addition, field trials have been approved for GMO maize, soy and cotton varieties.
Genetically modified brinjal (eggplant) is Bangladesh’s first genetically modified crop that protects brinjal from the deadly fruit and shoot borer. The release of the insect resistant eggplant varieties made Bangladesh the first developing nation to release a genetically engineered commercialized food crop. Cultivation in 2019 of the insect-resistant brinjal increased 28-fold over 2018 to 4770 acres. The insect-resistant trait helped to save farmers $120 per hectare (2.7 acres) by significantly lowering pesticide applications by 70 to 90 percent and increasing yield by 30 percent. The estimated economic benefit per hectare is over $1800. There is ongoing research and field trials for new varieties of insect-resistant brinjal. The Bangladesh Agricultural Research Institute is also working to develop GM potato, rice, cotton and tomato.
GMO Crops in Canada and the United States
Canada and the United States are two of the original six countries to begin planting GM crops in 1996. (The other four are Argentina, Australia, China and Mexico.) Since these first plantings, up to 10 biotech crops have been produced on hundreds of million acres across both nations, with the cumulative economic benefit from biotech crops netted between 1996 and 2018 over $105.6 billion.
Among the 26 countries growing GM crops in 2016, Canada ranks fourth in production, cultivating canola, corn, soy, sugar beets, alfalfa and potatoes on 31 million acres. Farmer adoption of the top three crops – canola, corn and soy - is 95 percent, 90 percent, and 82 percent, respectively. CropLife Canada estimates that the use of biotech crops and pesticides has enabled farmers to produce 42 percent more grain worth an added CA$ 8.3 billion on an annual basis.
Originally developed in Canada in the 1960s and 70s, canola (an abbreviation of “Canadian oil”) is a variety of rapeseed in which the erucic acid and glucosinolate content must be below specified levels. Erucic acid, though not a safety concern at normal consumption rates, has been shown to have toxic effects at high enough levels. Canola oil is used in a wide variety of food and industrial products, such as salad oil, shortening, spreads and creamers, as well as printing ink, plastics and cosmetics. Canada, fittingly, was the first country to commercialize herbicide-tolerant canola in 1996 and exports 90 percent of its canola crop to China, Japan, Mexico and the U.S.
To date, Canada has approved 185 biotech traits for food, feed and cultivation, including the non-browning/low-bruising potato and the non-browning apple, which was developed by Okanagan Specialty Fruit (OSF) based in British Columbia, Canada. Since 2017, farmers have been growing GM potatoes, and although not currently grown, three biotech apples have been approved for cultivation in Canada.
The United States is the largest producer of genetically modified crops in the world, planting 38 percent of total global production. In 2019 farmers in the U.S. grew GM corn, cotton, soybeans, canola, sugar beets, alfalfa, papaya, potatoes, apples and summer squash on over 175 million acres. In the 20 years of commercialization of biotech crops (1996-2015), the United States accrued the highest benefits at $72.9 billion and $6.9 billion for 2015 alone. Farmer adoption rates for the major biotech commodity crops in 2016 were 92 percent for corn, 94 percent for soybeans, 98 percent for cotton, and 100 percent for canola and sugar beets.
According to USDA the U.S. is the world’s largest agricultural exporter, and a majority of the corn and soy products exported abroad are biotech varieties. Both the EU and China are major markets for U.S. grain exports, with an estimated 6.5 million tons of soybeans exported to the EU. The United States Department of Agriculture recently approved a drought tolerant soybean, HB4, that has also been approved for cultivation in Brazil and Argentina.
Like Canada, two of the most recently approved crops are the GM non-browning and low-bruising Innate® potato and the non-browning Arctic® apple. These two crops were developed to encourage less food waste from the farm to the kitchen table. It is estimated that approximately 133 billion pounds of food is wasted annually in the U.S., much of that waste from discarded fruits and vegetables.
In 2016 farmers planted over 4400 acres of the GM Innate® potato. This biotech potato also has reduced amounts of asparagine, which when cooked at high temperatures, forms acrylamide, a potential carcinogen. A second generation Innate® potato with the same traits as the first generation plus late blight resistance and reduced sugars also has been grown by US farmers in 2019. Late blight is the fungal disease that led to the Great Irish Famine in the 19th century and still affects farmers around the world today.
The biotech Arctic® apple varieties were first commercially harvested in 2016 and is now available for sale in the U.S. The Arctic® apple’s non-browning trait ensures the apple will not brown if it is cut, bitten or bruised, encouraging growers, packers, retailers and consumers alike to waste less fruit due to superficial discoloration.
Scientists are eager about the potential for non-browning transgenic plants to serve as a possible solution towards the food waste situation. In 2019, researchers successfully inserted the non-browning gene into romaine lettuce. The product has been given “deregulated” status by the United States Department of Agriculture’s Animal and Plant Health Inspection Service and will hopefully be commercialized and on the market in the next 2 years.
Field trials are currently being conducted on two other tree species, both under attack from diseases. In the early 20th century, the American chestnut was devastated by chestnut blight, a fungal disease that eradicated the hardwood tree from East Coast forests. Researchers at SUNY-ESF have been working to develop a blight-resistant American chestnut, incorporating resistance genes from the Chinese chestnut. The research team has launched the 10,000 Chestnut Challenge, with the goal to plant 10,000 blight resistant trees over the scope of a 5-year period.
Citrus greening, another plant disease, is decimating Florida’s $5.1 billion citrus industry. Citrus greening is a brutal disease, reducing the production volume of Florida’s citrus industry by 74% since it entered the state in 2005. Texas A&M University scientists, with support from Southern Gardens, have been developing a disease-resistant citrus tree that utilizes a gene from spinach to confer resistance. Following successful field trials, Southern Gardens is looking to gain approval for the disease-resistant tree that could be available for planting in three to four years.
Canada and the U.S. have approved the AquaBounty salmon. The AquaBounty salmon grows to market size in half the time of wild-type salmon. Regulatory agencies in both countries have confirmed the GM salmon to be as safe and nutritious as non-GM salmon already on the market. After delays caused by the COVID-19 pandemic, the first AquaBounty salmon has officially been sold to a few restaurants and distributors in the United States in mid-2021. The United States Food and Drug Administration has approved the import and commercialization of GM salmon eggs from Canada.
GMO Crops in Europe
In 2010 the European Commission (EC) published an extensive report highlighting data gleaned from over two decades of public research funded by the European Union on genetically modified crops. According to the EC, “The main conclusion to be drawn from the efforts of more than 130 research projects, covering a period of more than 25 years of research, and involving more than 500 independent research groups, is that biotechnology, and in particular GMOs, are not per se more risky than e.g. conventional plant breeding technologies.”
Despite the confirmed safety of biotech crops by the EC, European Academy of Sciences, and the European Food Safety Authority (EFSA), public anti-GMO sentiment remains prevalent throughout the EU, impacting the rate of approvals for new GM crops and farmers’ access to biotech seeds. The EU, however, continues to import vast amounts of GM grains for livestock, and farmers in Spain and Portugal grow GM maize. The United Kingdom (UK) is steadily moving closer to cultivation, conducting research and development on GM crop varieties, in addition to receiving support from outspoken public figures.
In the EU, all GM crops for importation or cultivation undergo a thorough risk assessment by EFSA. To date, EFSA has issued positive scientific opinions on 109 events and consistently confirmed the safety of all GM crops on the market. In the EU, member states vote on an individual basis regarding domestic approvals for cultivating biotech crops. Currently, 19 of the 25 EU countries voted to opt out of cultivating biotech crops. Despite strict bans of domestic production, the EU remains one of the largest importers of biotech grains for livestock feed and industrial uses. CropLife Europe has found that there is no correlation between EU member state approval voting patterns and quantities of GM grain imported per capita by that member state, meaning that some nations who vote for GM cultivation bans are also some of the nations importing the most GM crops.
Despite limited cultivation, the EU GM grain imports are very high value EU livestock farmers rely on imports for 70 percent of their grain needs, including up to 34 million tons of GM soy. CropLife Europe notes that stopping imports of GM feed to Europe would result in 30 billion euros in economic losses annually.
Currently, two European countries – Spain and Portugal –plant genetically modified maize, nearly 276,000 acres in 2019, where it is used primarily in livestock feed. This represents a declining area, as acreage of GM crops in the EU has dropped by over 7% since 2018.
With the UK having officially transitioned out of membership in the EU, the national government will have the responsibility in creating new agricultural trade and production policy on their own. Although the UK has yet to approve a biotech crop for cultivation, the UK is leading Europe in innovative agriculture biotechnology research, and government and other public figures continue to speak out in support of GM crops. In 2021, the Minister of Farming and Environment for England has identified England’s priority to reevaluate the regulatory process regarding various agrobiotechnology techniques as compared to conventional breeding strategies, with the hopes of simplifying the GM crop approval process.
The United Kingdom’s Rothamsted Research is the oldest running agricultural research station in the world. Rothamsted scientists have been working to develop and run field test-trials for a genetically engineered wheat and camelina varieties modified using CRISPR-Cas9 technology to reduce the amount of asparagine in the wheat grain. Asparagine is one of the 20 common amino acids used in protein synthesis, but excess levels of asparagine in the flour can lead to the formation of a toxic compound, acrylamide, during baking. This project is a part of a multi-institutional research initiative across 8 research centers in the UK called Designing Future Wheat.
Additionally, the John Innes Center, a leading plant science research center in Norwich, UK has joined the Alliance for Accelerated Crop Improvement in Africa (ACACIA). The objectives of this alliance include accelerating crop improvement in Africa by developing resources, research capacity, and key partnerships. One of the current research projects UK scientists are engaged on in this project regards breeding grasspea, a legume plant with potential to grow in harsh climates while producing protein and improving soil health, for cultivation all across sub-Saharan Africa. Wild grasspea contains some toxic secondary metabolites, so researchers are trying to determine ways to make the crop palatable for human consumption.
GMO Crops in Latin America
For more detailed and up-to-date information on GMO crops in Latin America, please visit the Biotec-LatAm website.
As a block Latin American countries are powerhouse producers of GM crops, with 10 countries growing over 207 million acres of maize, soybeans, cotton and canola in 2016. Brazil leads Latin American nations in cultivation and exports, followed by Argentina. Paraguay, Bolivia, Uruguay, Mexico, Colombia, Chile, Honduras, and Costa Rica too grow GM varieties. Latin American countries also are biotechnology innovators, developing local GM crops, often as an outcome of public-private partnerships.
Since 1998 Brazil has approved 111 biotech traits, and ISAAA cites that economic benefits to farmers planting biotech crops between 2003 and 2018 are $26.6 billion, with $3.4 billion gained in 2018 along. In 2016 Brazil grew over 87 million acres of GM soybeans, maize and cotton and is a leading exporter in all three crops. Both China and the European Union import biotech cotton and soybeans from Brazil, and GM maize is exported from Brazil to Iran and Asian countries.
Brazil has approved a biotech higher-yield eucalyptus (the first approval for eucalyptus worldwide) for bioindustrial usage, as well as a GM virus-resistant bean and an herbicide-tolerant soybean. Private sector companies, BASF and the Center of Technology in Sugarcane, working with the public institution EMBRAPA, the Brazilian Agricultural Research Corporation, have been developing and testing an herbicide-tolerant soybean and drought-tolerant sugarcane, respectively. Oxitec, in partnership with the University of Sao Paolo, commercialized a genetically engineered mosquito that is modified to help fight devastating tropical diseases, such as Zika virus and Dengue, Yellow, and Chikungunya Fever. In recent years, Oxitec has released genetically modified agricultural insect pests, that produce progeny with a gene that prevents them from surviving to adulthood.
Second after Brazil, Argentina has approved 78 biotech traits and produced nearly 59 million acres of GM maize, soybeans, cotton, and alfalfa (for the first time) in 2019. Over the 20 years of planting, GMO production has increased Argentina farm income by approximately over $28 billion. Argentina is the largest producer of soybean meal globally, with nearly 100 percent of their crop biotech soybeans, and exporting nearly 11 MMT. In 2019, Argentina approved many new traits, including 6 HT/IR stacked maize traits. Additionally, this was the first year Argentine farmers grew low-lignin, herbicide tolerant, and stacked event alfalfa. Alfalfa is the most important forage crop in Argentina, as it is often used for grazing for both beef and dairy herds in the country. Beef and dairy are growing industries in Argentina, so there is large potential that GM alfalfa could follow similar growth trends in subsequent years.
Eight other Latin American countries - Bolivia, Chile, Colombia, Costa Rica, Honduras, Mexico, Uruguay and Paraguay – also cultivate GM food and horticulture crops, including maize, cotton, canola, soybeans, carnations and roses.
GM soybean cultivation in Bolivia has grown to nearly 3.5 million acres, elevating Bolivia as the ninth largest soybean producer in the world and increasing farmer economic benefits from 2008 to 2018 by $874 million. Bolivia’s crop is exported to other Latin American countries, including Ecuador, Peru and Venezuela. Currently on glyphosate tolerant soybeans have been approved for cultivation, but farmers are pushing for approvals of drought tolerant seed, as rainfall is often the most limiting factor in Bolivian crop production.
Currently, Chile grows over 100,000 acres of GM maize, canola and soybeans only for seed for export. Chile ranks ninth worldwide in overall seed production and first in GM seed production, with most of its GM seeds exported to the United States and Canada. Despite the prohibition to grow GM crops for internal consumption, Chile imports GM grains for livestock feed and processed food products containing GM ingredients.
There are several public research projects in Chile to develop genetically engineered varieties of local crops. A researcher from the University of Talca has developed a transgenic strain of maize that is extremely drought-tolerant. The new strain can survive for 52 days without water while maintaining approximately 80% productivity. This innovation, if approved, could strongly benefit farmers in a country whose landscape is dominated by the dry regions surrounding the Atacama Desert.
Colombia produced over 250,000 acres of GM maize and cotton in 2016, and since GM plantings began in 2004, farm income has increased by $302 million. Colombia also grows genetically engineered Dutch blue carnations for export to Europe and GM blue petal roses for export to Japan, where one rose can retail for up to $50. There are numerous ongoing public and private research projects to develop genetically engineered varieties of cassava, coffee, rice, potatoes and sugarcane.
Similar to Chile, Costa Rica also grows biotech crops for seed production to export to Canada and the United States. In 2019, Costa Rica grew over 730 acres of biotech cotton, soybeans, and uniquely, a GM pineapple with high antioxidants. In addition local scientists are developing and testing an herbicide-tolerant rice and GM bananas that are resistant to black Sigatoka, a plant disease that can lead to 50 percent or more yield loss.
Honduras increased its GMO maize cultivation in 2019 by 5 percent over 2018, cultivating more than 92,000 acres. Honduras grows GM maize for animal feed, human consumption, and for seed, exporting GM maize seed to Colombia. It is estimated that since 2002, farm income has been enhanced by biotech crops by $20.9. million. Honduras also imports GM feed for their livestock and aquaculture industries.
In 2019 8,000 Mexican farmers cultivated nearly 590,000 acres of GM cotton and to date Mexico has authorized 188 events for import and consumption, including soybeans, maize, rice, alfalfa, canola, potatoes, tomatoes, and sugar beets. From 1996 to 2018, Mexican farmers have gained $798.5 million in income by planting biotech crops. The International Maize and Wheat Improvement Center (CYMMYT) based in Mexico has been testing genetically engineered wheat, and new facilities partially funded by the Bill and Melinda Gates Foundation will enable further research in GM maize and wheat.
Paraguay is the sixth largest producer of biotech crops globally. Paraguay grew over 10 million acres of GM cotton, maize and soybeans in 2019. Paraguay is the third largest biotech producer in Latin America, exporting to Brazil, the EU, Israel and Russia. Nearly 99 percent of the soybean crop is biotech. Since original plantings in 2004, Paraguay farm income has increased by $2.3 billion.
Finally, Uruguay is the eleventh largest producer of biotech crops globally, growing nearly three million acres of GM maize and soybeans, with 97 percent adoption of biotech soybeans. Over the 15 years Uruguay’s farmers have planted biotech crops, their income has increased by $214 million. Uruguay exports its soybeans to Egypt, the EU, China and other Asian countries.